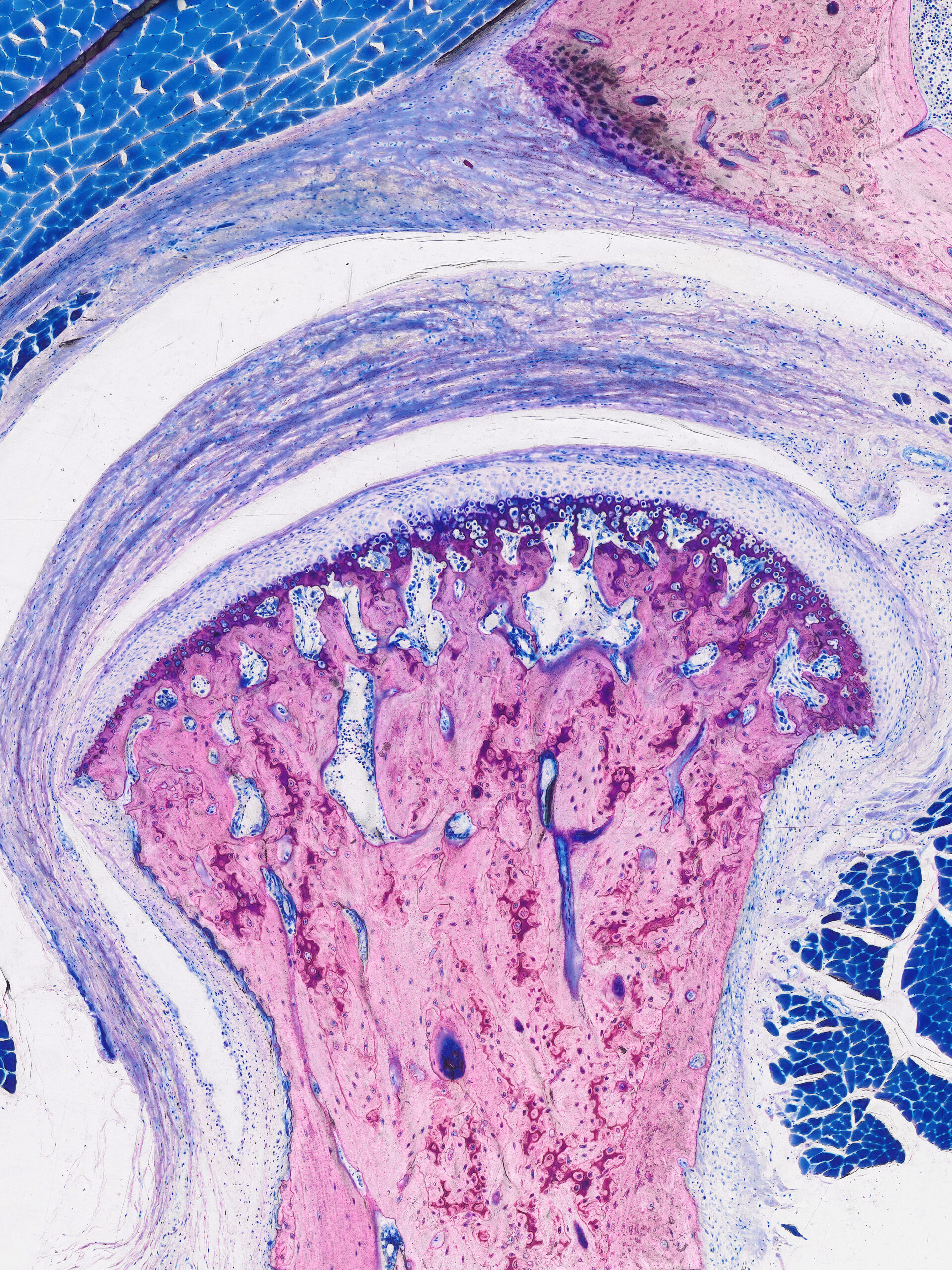
Injuries of bones are a frequent consequence of trauma, such as traffic accidents, osteoporosis-related fractures and sports injuries. A quick and full recovery of these injuries is of great importance. In some cases, the treatment is especially challenging due to the compromised healing and regenerative potential of the tissue, resulting either from damage to blood supply, systemic conditions or advanced patient age. In order to reach successful healing of such injuries, surgical intervention and tissue grafting are often required.
The group lead by Regina Brunauer, under the clinical-co lead of Thomas Nau, is developing new approaches to improve bone regeneration. The overall focus is on the development of tissue replacement materials and biological tissue substitutes with enhanced functionality. The research areas encompass:
- Novel osteosynthesis materials and bone substitutes, involving combinations of scaffolds, growth factors, stem cells and pre-vascularization approaches
- Advanced non-viral gene therapy approaches for bone regeneration
- Approaches for bone tissue regeneration in elderly patients with compromised native regenerative potential
- Novel biomarkers associated with increased risk for bone injury and degenerative joint diseases
The approaches are examined regarding their efficiency and ethical justifiability in practice. Working in close collaboration with clinicians, a particular focus is placed on the strategies supporting translation into practice.